Abstract
Background: Central nervous system (CNS) relapse (CNSr) in patients with aggressive non-Hodgkin lymphoma is uncommon but carries a high morbidity and mortality. Data in T- and NK-cell lymphomas (TCL) are sparse, though the few small case series describing CNSr in TCL report an incidence up to 9% with median overall survival (mOS) less than 3 months (mo). We previously reported a large (n=75) case series of CNSr in TCL (Abstract #1382, ASH 2021). Herein, we developed a prediction score for CNSr in newly diagnosed patients with TCL.
Methods: We retrospectively collected clinicopathologic and treatment data from 19 US academic centers from patients diagnosed with TCL between 1/1/09-1/1/19 who experienced CNSr (CNSr cohort). We combined these data with that from patients diagnosed with TCL without CNSr from a single institution (Penn cohort) to create a training set (T-set) enriched for patients with CNSr. Patients with leukemic or cutaneous TCL subtypes were excluded. We applied a LASSO Cox proportional hazards model to the T-set to create a predictive score for factors associated with CNSr and validated this score in an independent population-based validation set (V-set) from the Swedish Lymphoma Registry, which was not enriched for CNSr.
Results: We evaluated data from 91 and 135 patients in the CNSr and Penn cohorts, respectively, which were combined to form the T-set and compared them to the V-set (n=745). Median age in the T-set was lower than in the V-set (60 vs 67; p<0.001). The most common TCL subtypes in the T-set and V-set were peripheral TCL, not otherwise specified (PTCL, NOS; 23% vs 34%, respectively; p=0.002), TCL with T-follicular helper phenotype (23% vs 14%, respectively; p=0.001), and ALK-negative anaplastic large cell lymphoma (18% vs 15%, respectively; p=0.352). CNS prophylaxis, defined as intrathecal therapy or high-dose methotrexate, was more common in the V-set than in the T-set (15% vs 9%, respectively; p=0.012). Rates of stem cell transplantation (SCT) as consolidation in first remission (CR1) did not differ significantly between the T-set and V-set (p=0.140). The median follow-up time was 37.6 mo in the T-set and 95.9 mo in the V-set. The median progression free survival (mPFS) and mOS after diagnosis were 9.8 mo and 33.2 mo in the T-set, respectively, and 8.5 mo and 17.9 mo in the V set, respectively. Within the T-set, mPFS and mOS were shorter for patients in the CNSr cohort vs the Penn cohort (6.0 mo vs 37.1 mo [p<0.001] and 17.3 mo vs 78.1 mo [p<0.001], respectively). In univariate analysis of the CNSr-enriched T-set, CNS prophylaxis, SCT in CR1, or frontline etoposide did not significantly decrease the risk of CNSr.
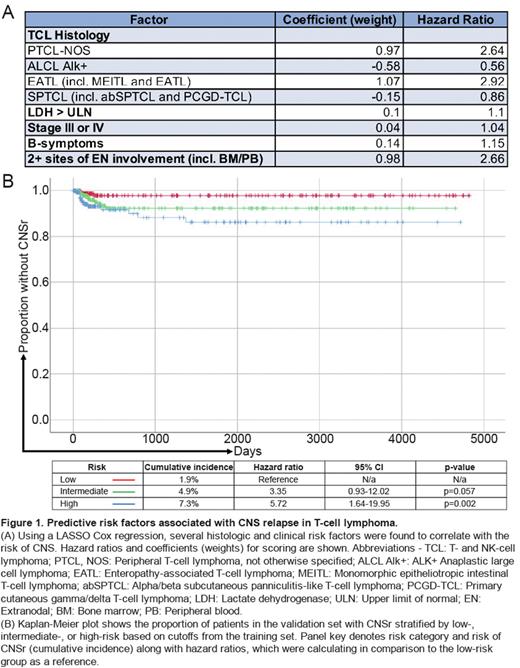
Using several clinicopathologic characteristics, we fitted a LASSO Cox proportional hazards model to the T-set, which selected histology, LDH, stage, B-symptoms, and ≥2 sites of extranodal involvement (ENI) for a weighted risk score of CNSr (Fig. 1A). Variables most strongly associated with CNSr were PTCL, NOS (HR 2.64), enteropathy-associated TCL (HR 2.92), and ≥2 sites of ENI (HR 2.66). We stratified the T-set into tertiles based on weighted scores (x), which was low-risk if x≤0.14, intermediate-risk if 0.14<x ≤1.11, or high-risk if x>1.11. Cumulative incidence of CNSr in each respective group was 19.6%, 55.1%, and 83.7%. When the same cutoffs were applied to weighted scores in the V-set, we confirmed separation into distinct low-, intermediate-, and high-risk groups with cumulative incidences of CNSr of 1.9%, 4.9%, and 7.3%, respectively (Fig. 1B). As the V-set population was not enriched for CNSr, the relative incidence is expectedly lower compared to the T-set. Compared to the low-risk group in the V-set, risk of CNSr was significantly increased in the high-risk group (HR 5.72, p=0.002) and increased with a trend toward significance in the intermediate-risk group (HR 3.35, p=0.057).
Conclusions: In a cohort of patients with TCL enriched for CNSr, we were able to characterize several risk factors associated with CNSr and developed a predictive CNSr in T-cell lymphoma Index (CITI) that could stratify patients into 3 distinct risk groups. PTCL, NOS, EATL, and ≥2 sites of ENI had the highest impact on risk of CNSr. The CITI score was validated in an independent population-based cohort. Although CNSr was uncommon even in high-risk patients (7.3%), our data will inform clinical decision making and may allow for identification of patients with TCL at high-risk of CNSr in future studies.
Disclosures
Horwitz:Affimed,: Consultancy; Yingli Pharma Limited and Tubulis: Honoraria; Takeda: Consultancy; Celgene: Research Funding; Affimed: Research Funding; ADC Therapeutics: Research Funding; SecuraBio: Honoraria; Shoreline Biosciences, Inc.: Membership on an entity's Board of Directors or advisory committees; Millennium /Takeda: Research Funding; Seattle Genetics,: Research Funding; C4: Research Funding; Verastem/SecuraBio: Research Funding; Kyowa Hakko Kirin: Research Funding; Daiichi Sankyo: Research Funding; Crispr Therapeutics: Research Funding; ONO Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Kyowa Hakko Kirin: Consultancy; Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees; Cimieo Therapeutics: Honoraria. Chavez:MorphoSys/Incyte: Speakers Bureau; ADC Therapeutics: Research Funding; Kite Pharma: Consultancy; GenMab: Consultancy; Epizyme: Honoraria, Speakers Bureau; Abbvie: Consultancy; Janssen: Research Funding; Merck: Research Funding; Astrazeneca: Research Funding, Speakers Bureau; Beigene: Honoraria; TG Therapeutics: Honoraria; Adicet: Consultancy. Sokol:Dren Bio: Consultancy; Kyowa-Kirin: Honoraria, Research Funding. Saeed:Novartis: Consultancy; Epizyme: Consultancy; Morphosys: Honoraria. Porcu:Teva: Honoraria, Research Funding; ADCT: Membership on an entity's Board of Directors or advisory committees; Loxo: Membership on an entity's Board of Directors or advisory committees; Ono: Membership on an entity's Board of Directors or advisory committees; DrenBio: Consultancy; BeiGene: Membership on an entity's Board of Directors or advisory committees; Innate Pharma: Honoraria, Membership on an entity's Board of Directors or advisory committees; Daiichi, Kyowa: Honoraria, Membership on an entity's Board of Directors or advisory committees; Morphosys: Membership on an entity's Board of Directors or advisory committees; Viracta: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Mehta-Shah:Corvus Pharmaceuticals: Research Funding; Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genetech/Roch: Membership on an entity's Board of Directors or advisory committees, Research Funding; Karyopharm Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Kyowa Hakko Kirin Co., Ltd.: Membership on an entity's Board of Directors or advisory committees; Secura Bio: Membership on an entity's Board of Directors or advisory committees, Research Funding; Verastem: Research Funding; Celgene: Research Funding; Bristol Myers-Squibb: Research Funding; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees. Zain:Secura Bio: Consultancy, Research Funding; Daichi Sankyo: Consultancy, Research Funding; AstraZeneca: Research Funding; Myeloid: Consultancy, Research Funding; CRSPR: Research Funding; Seattle Genetics: Research Funding, Speakers Bureau; 3M: Current holder of stock options in a privately-held company; Affirmed: Research Funding; Kiyowa Kirin: Speakers Bureau. Brammer:DrenBio: Consultancy; Kymera Therapeutics: Consultancy; Seattle Genetics: Speakers Bureau; Bristol-Myers Squibb: Research Funding. Iyer:CureBio: Honoraria; Salarius Pharmaceuticals, Inc.: Consultancy; Rhizen: Research Funding; Spectrum: Research Funding; Innate: Research Funding; Merck: Research Funding; Legend: Research Funding; Takeda: Research Funding; Affimed: Research Funding; Seagen: Consultancy, Research Funding; Yingli: Consultancy, Research Funding; CRISPR Therapeutics: Research Funding; Target Oncology: Consultancy, Honoraria; Myeloid: Research Funding. Olszewski:Genentech: Research Funding; Genmab: Consultancy, Research Funding; Precision Bio: Research Funding; Adaptive Biotechnologies: Research Funding; Celldex: Research Funding; Acrotech Biopharma: Research Funding; Schrodinger: Consultancy; TG Therapeutics: Consultancy, Research Funding. Smith:TGTX: Consultancy; Bayer: Consultancy; Genentech: Consultancy; Kite Pharma: Consultancy; ADC Therapeutics: Consultancy; Gilead: Consultancy; BMS: Consultancy; Morphosys: Consultancy; Adaptive: Consultancy; Janssen: Consultancy; Karyopharm: Consultancy; Celgene: Consultancy; Portola: Research Funding; Gamida Cell: Consultancy; Bantam: Consultancy; Karyopharm: Consultancy; Chair, Lymphoma Research Foundation SAB: Membership on an entity's Board of Directors or advisory committees. Riedell:Sana Biotechnology: Consultancy; Xencor: Research Funding; MorphoSys: Research Funding; Tessa Therapeutics: Research Funding; BeiGene: Consultancy; Takeda: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Intellia Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees; Calibr: Research Funding; Kite/Gilead: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Nurix Therapeutics: Membership on an entity's Board of Directors or advisory committees; Nektar Therapeutics: Membership on an entity's Board of Directors or advisory committees; Fate Therapeutics: Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees. Haverkos:Viracta Therapeutics: Consultancy; Bristol Myers Squibb: Research Funding. Hu:Genentech/Roche: Research Funding; Janssen Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Incyte: Membership on an entity's Board of Directors or advisory committees; Beigene: Membership on an entity's Board of Directors or advisory committees, Research Funding. Allen:Daiichi Sanyko: Consultancy, Honoraria; Kyowa: Consultancy, Honoraria. Jagadeesh:Debio pharma: Research Funding; LOXO Pharmaceuticals: Research Funding; MEI Pharma: Research Funding; Regeneron Pharmaceuticals, Inc.: Research Funding; Seagen: Research Funding; Daiichi Sankyo: Consultancy, Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Research Funding; ATARA Biotherapeutics: Research Funding; Affimed: Membership on an entity's Board of Directors or advisory committees; Trillium Pharmaceuticals: Research Funding. Ghione:Kite Pharma: Research Funding; Secura Bio: Consultancy; Kyowa Hakko Kirin: Consultancy; AstraZeneca Pharmaceuticals: Consultancy. Rhodes:Velosbios: Research Funding; SeaGen: Consultancy; Pharmacyclics: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Genmab: Consultancy; Abbive: Consultancy; Verastem: Consultancy; Morphosys: Consultancy; Genentech: Consultancy; TG Therapeutics: Consultancy; Beigene: Consultancy; Oncternal: Research Funding; Loxo Oncology: Research Funding; Epizyme: Research Funding. Chong:Juno/BMS: Consultancy; Novartis: Consultancy; Beigene: Consultancy; Tessa: Consultancy; KITE: Consultancy. Gerson:Abbvie: Consultancy; Loxo Oncology: Research Funding; Genentech: Consultancy. Landsburg:Triphase: Research Funding; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Morphosys: Membership on an entity's Board of Directors or advisory committees; Calithera: Membership on an entity's Board of Directors or advisory committees; Curis, Inc: Research Funding; Epizyme: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees. Dwivedy Nasta:Roche: Research Funding; Rafael: Research Funding; Pharmacyclics: Research Funding; FortySeven/Gilead: Research Funding. Schuster:DTRM: Research Funding; Celgene: Consultancy, Honoraria, Research Funding; AbbVie: Research Funding; Adaptive Biotechnologies: Research Funding; Pharmacyclics: Research Funding; TG Therapeutics: Research Funding; AstraZeneca: Consultancy; Fate Therapeutics: Consultancy; Legend Biotech: Consultancy; Loxo Oncology: Consultancy; Mustang Biotech: Consultancy; Morphosys: Consultancy; Incyte: Consultancy, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Merck: Research Funding; Juno Therapeutics: Consultancy, Research Funding; Roche: Consultancy, Research Funding; Genmab: Consultancy; BeiGene: Consultancy; Genentech: Consultancy, Research Funding; Nordic Nanovector: Consultancy; Regeneron: Consultancy; N/A: Patents & Royalties: Related to CD19 CAR T cells and autologous co-stimulated T cells. Svoboda:TG: Research Funding; SEAGEN: Consultancy, Research Funding; Pharmacyclics: Consultancy, Research Funding; Merck: Research Funding; Incyte: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genmab: Consultancy; BMS: Consultancy, Research Funding; Atara: Consultancy; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees, Research Funding; Adaptive: Membership on an entity's Board of Directors or advisory committees, Research Funding; ADCT: Consultancy. Jerkeman:Incyte: Honoraria; Kite/Gilead: Consultancy, Honoraria, Research Funding; Orion: Honoraria; Novartis: Honoraria; Genmab: Honoraria; AstraZeneca: Honoraria, Research Funding; Roche: Honoraria, Research Funding; Janssen: Honoraria, Research Funding; BMS: Honoraria, Research Funding; Abbvie: Honoraria, Research Funding. Barta:Seagen: Honoraria; Kyowa Kirin: Consultancy, Honoraria; Daiichi Sankyo: Consultancy; Affimed: Consultancy; Janssen: Other: Independent Data Monitoring Committee member; Acrotech: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal